AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Copper pourbaix diagram1/2/2023  
It also indicates the need to minimize accumulation of copper and iron oxides and of acid-forming salts in the steam generators.Īuthors: Daniel, P L Harper, S L Publication Date: Wed Oct 01 00:00: Research Org.: Babcock and Wilcox Co., Alliance, OH (USA). This work corroborates the postulated pitting mechanism and suggests a mechanism of formation for the observed wastage bands. 
Local solute concentrations of 0.001 to 0.1 molar are consistent with the postulated pH. Pitting occurs in areas where reduction of copper oxides raises the Alloy 600 surface potential into the passive range. The wastage band near the tubesheet is attributed to active dissolution of Alloy 600, which occurs at low potentials. Indications are that the pH within the pits and wastage areas ranges from 1 to 4 - the higher pH occurs at the tube surface and in pits near the tubesheet the lower pH occurs in pits away from the tubesheet. They were used to analyze local corrosive conditions and corrosion processes that occur in the steam generators. The diagrams were constructed using new thermodynamic data. įIGURE 56.7 Pourbaix diagram of the copper- chlorine-water system.Corrosion products identified on Alloy 600 tubes pulled from the Millstone Unit 2 nuclear steam generator were compared to phase fields of appropriate Pourbaix diagrams. The Pourbaix diagrams relate to 25 ☌ but, as is known, it is often necessary to implement operation at elevated temperatures to improve reaction rates, and at elevated temperatures used in practice the thermodynamic equilibria calculated at 25 ☌ are no longer valid. Thus, the potential-pH diagrams for simple metal- water systems are not directly applicable in these cases. For example, in ammoniacal solutions, nickel, cobalt, and copper are present as complex ions which are characterized by their different stabilities from hydrated ions. One factor which has an important bearing on the thermodynamics of metal ions in aqueous solutions is the presence of complex ions. It is important to recognize some of the limitations of the Pourbaix diagrams. įigure 2.22 Pourbaix diagram for the copper-water system at 25° C. The oxide of copper, Cu+ and Cu " " are only. Copper is not passive in acid electrolytes. Copper (E° = 0.337 V) is more noble than iron ( p = -0.444V), however, it is more stable in water (SHE) than iron. Ī Pourbaix diagram for copper/water system is shown in Fig. įig- 7.36 Pourbaix diagram for the system copper-water. This greater nobility results in copper being thermodynamically stable in water that is, line 14 (-6) representing aCu2+ = 10-6 lies above line a. 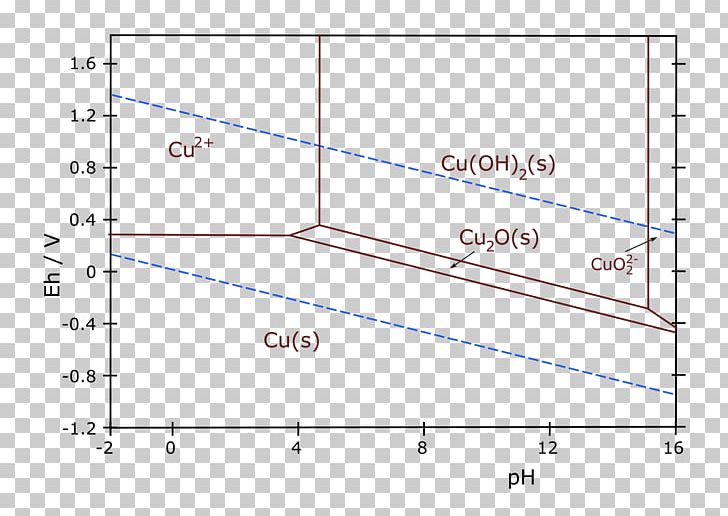
The more positive standard electrode potential of copper (+337 mV (SHE)) as compared to iron (-440 mV (SHE)) is evident. The Pourbaix diagram for the copper/water system is shown in Fig. FIGURE 7.1 Pourbaix diagram of copper-water system (from Ref. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
